Medical
About Us


Sustainability
Medical
More and more diseases are becoming susceptible to medical treatment today, raising life expectancies all over the world, with preventive medicine and early diagnosis taking on increasingly important roles in sustaining long, healthy lives. At the Eiken Group, we view promoting the adoption of these two approaches as vital to the search for solutions to medical issues. The diagnostics offered by the Eiken Group already play a familiar part in the medical lives of millions, with applications ranging from prenatal gynecological examinations, examination of newborn babies for metabolic abnormalities and health checkups for children to cancer screening and screening for lifestyle diseases. As a pioneer in clinical diagnostics, the Eiken Group is proud to develop and circulate products and technologies serving advanced medical needs. Through these efforts, the Group is determined to contribute to the development of medicine and achievement of healthy lives for people everywhere while capturing new business opportunities and attaining sustainable growth.
Eiken Chemical adopted the immunochemical method (latex agglutination method) for its fecal immunochemical test (FIT) reagents in 1987 and also developed the world’s first fully-automated FIT analyzer in 1989. We have achieved top market share in Japan and received recognition for the performance and knowledge backing our products all over the world. They are now used in colorectal cancer screening programs in many countries. The goal of cancer screenings is early detection of the disease for early treatment which reduces mortality rates. Taking FIT regularly enables early detection of colorectal cancer and has a scientifically-based potential to reduce mortality rates. When colorectal cancer is detected and treated in the early stages, the 5-year survival rate is over 90%, and the possibility of recovery is high. The colorectal cancer screening programs using our reagents and analyzers contribute to global early detection of this disease.
In addition, we sell reagents for “stratified gastric cancer risk assessment,” blood testing to investigate Helicobacter pylori infection and stomach lining atrophy factors which can increase the likelihood of future gastric cancer development, and we are working to familiarize this testing method inside and outside of Japan.


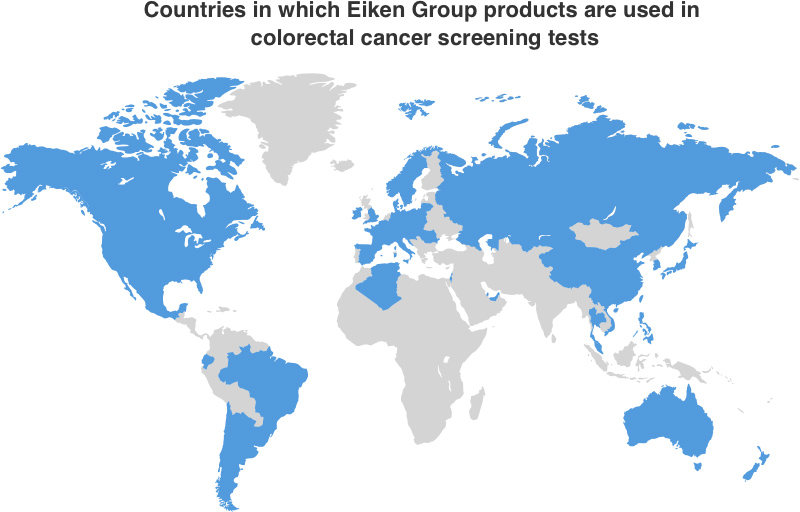
| Results for FY2024 | Target for FY2027 |
|
|||
|---|---|---|---|---|---|
| Number of countries in which colorectal cancer screening tests are | 52 | 61 | 67 |
To contribute to the solution of medical issues globally, the Eiken Group must constantly change and advance, never clinging to the conventional boundaries of clinical testing. Through open innovation in partnership with government, academia and other industrial companies, the Eiken Group is developing companion diagnostic drugs for individually tailored treatments, creating POCT platforms for the primary care field, and advancing into new fields such as risk diagnostics and wearable health devices. In this way the Group is pursuing new approaches in diagnostic testing to support the health of people worldwide. In 2021 the Group concluded an agreement with Illumina, Inc. of the United States to use that company’s next-generation sequencer, the Miseq™ Dx system, to advance the development of companion diagnostic systems. Going forward, the Eiken Group will continue its vigorous pursuit of solutions to medical problems, including through partnerships such as these.
The dysentery that swept Japan after the war. Severe Acute Respiratory Syndrome, which emerged in 2003. The new strain of influenza of 2009.
The mission of the Eiken Group is “to safeguard the health of people worldwide.” Since its establishment in 1939, the Eiken Group has contributed to Japan’s public health and communicable-disease control by developing clinical testing reagents in partnership with medical practitioners. In 2020, when the COVID-19 pandemic swept the world, the Group quickly drew on its accumulated technology and experience to develop and sell genetic testing reagents. It did so using the LAMP method, a gene amplification technology developed exclusively by Eiken Chemical. To respond to heavy demand for clinical testing reagents, the Group is ramping up production capacity, striving to ensure stable supplies. Today, as infectious diseases continue to spread, the role of genetic testing is proving more crucial than ever. Economic development, global warming and rising mobility due to globalization make accurate diagnosis vital in the fight against not only COVID-19 but also a host of infectious diseases arriving from overseas. Mindful of its responsibilities as a producer of clinical testing reagents, the Eiken Group is developing not only testing reagents to tackle the infectious diseases of the future but also equipment and devices. In so doing, the Group is advancing the field of clinical testing, contributing to a society made resilient through the power of testing.


Since August 1955, Eiken Chemical has published Modern Media, a monthly academic journal. This periodical discusses topics related to human health from a wide array of perspectives, offering detailed yet accessible explanations by specialists in each field. Avid readers of Modern Media include subscribers at hospitals throughout Japan; at university medical, pharmacological and veterinary departments; at national, public and private research facilities; and among experts in diverse fields encompassing food, the environment, social services and more, for a circulation of 5,500 copies. In 2004, to share the journal’s academic information with a wider audience, Eiken Chemical launched the online version of Modern Media on Eiken Chemical website, with the same content as the printed journal. In 2020 the website was revamped, improving ease of use and adding new content. Modern Media will publish issue number 800 in July 2022.
Modern Media will continue to publish a wealth of valuable information on all things related to health.

Within three years of its launch, Modern Media‘s editorial office was gathering views and impressions from every corner of Japan. To establish a forum for two-way communication, the journal added a column entitled “Readers’ Column: From North, From South,” to showcase readers’ perspectives. Adorning this column was an illustration of a swallow, a bird that flies from one end of the Japanese archipelago to the other in spring. Although “From North, From South” was unfortunately discontinued, the policy of featuring the swallow continued, embodying the spirit of bringing readers together and as a symbolic messenger between the editorial office and readers. When Modern Media published its 500th issue, the swallow was adopted as its logo in commemoration. The seedling in the swallow’s mouth symbolizes the wide range of information and perspectives Modern Media brings to its readers, which may sprout and grow wherever it arrives into a great tree of knowledge.
Eiken Chemical sells books related to microbiological tests in clinical and food production settings. We hope these publications will be beneficial for students aiming to become clinical laboratory technicians in the future and for current working clinical laboratory technicians as well, assisting large numbers of people involved with the medical field. In addition, we also sell books incorporating basic knowledge which can be used to assist day-to-day inspections by workers involved with microbial testing of foodstuffs.
Eiken Chemical holds seminars related to essential aspects of testing such as basic knowledge, technology, and cautionary points targeted to workers involved with microbiological tests of foodstuffs. These seminars are divided into levels ranging from basic to intermediate and are designed to assist testing supervisors and workers involved with food hygiene in carrying out their jobs.
EIKEN CHEMICAL supports the activity of KUROZUMI MEDICAL FOUNDATION, which contributes to national health and improvement in welfare through subsidizing research on clinical diagnosis, hygiene tests and related basic medical sciences, as well as awarding research results.
This forum, which was founded with the goal of establishing effective urine testing (qualitative testing and sediment testing) for the fields of nephrology and urology, is engaged in mutual knowledge and information sharing activities for wide-ranging applications of non-invasive urine testing among doctors, clinical laboratory technicians, and clinical testing researchers. Eiken Chemical approves and supports these activities.

EIKEN support and support the purpose of the non-profit organization Brave Circle Steering Committee to expand the circle of screening for the promotion of understanding and early detection of colorectal cancer. This campaign is calling on the importance of colorectal cancer screening to many people through various activities in cooperation with governments, corporations, health insurance associations and other organizations.

Eiken Chemical is participating in “corporate action for cancer screening promotion,” a nationwide program which seeks to increase cancer screening rates, as a promotional partner company. These activities consist of companies taking the initiative to drive cancer screening rates to 50% and above by spreading information about their importance.
