Medical
About Us


About Eiken
1939
Company founded by Tsuyoshi Kurozumi and others as Koa Kagakukogyo Co., Ltd. (Katsushika-ku, Tokyo)
Launched manufacturing and sales of nutritional foods and medical supplies made with livestock organs as raw materials
1940
Company name changed to Koa Eiyokagaku Kenkyusho Co., Ltd.
1943
Exclusive distribution agreement and capital alliance executed with Tanabe Seiyaku Co., Ltd.
1946
Company name changed to Nihon Eiyokagaku Co., Ltd.
 Nihon Eiyokagaku NEK logo
Nihon Eiyokagaku NEK logo
 Nihon Eiyokagaku carbon logo
Nihon Eiyokagaku carbon logo
 Research laboratory interior
Research laboratory interior
 Research laboratory interior
Research laboratory interior
 Laboratory staff posed around Tsuyoshi Kurozumi, President
Laboratory staff posed around Tsuyoshi Kurozumi, President
1950
Launched sales of bacterial testing products
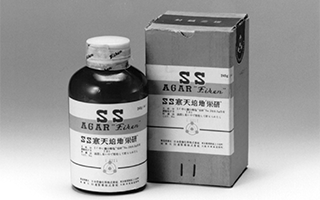 Powdered agar for the detection of bacteria (SS Agar "Eiken")
Powdered agar for the detection of bacteria (SS Agar "Eiken")
First company to succeed in creating a powdered agar for the detection of bacteria (isolation of typhoid and shigella bacteria), contributing to the development of communicable disease control and public health in Japan
1955
First published "Modern Media," a clinical diagnostics academic information magazine
 Modern Media August 1955 issue
Modern Media August 1955 issue
1958
Launched sales of bacterial drug sensitivity testing reagents (disk diffusion method)
Launched sales of tuberculosis culture medium "Ogawa Agar 'Eiken'"
1959
Founded EIKEN KIZAI CO., LTD.
1960
Completed new plant in Katsushika (Katsushika-ku, Tokyo: Katsushika Office)
Established the Clinical Diagnostics Division the following year and launched research and development of in vitro diagnostics
 Opened Nihon Eiyokagaku Plant 1 and Plant 2
Opened Nihon Eiyokagaku Plant 1 and Plant 2
1963
Launched sales of clinical chemistry reagents
 "Alkaline Phosphatase Tube 'Eiken'"
"Alkaline Phosphatase Tube 'Eiken'"
Launched sales of urinalysis test reagents
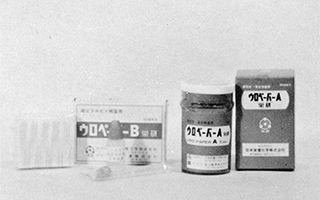 "Uropaper A 'Eiken'" and "Uropaper B 'Eiken'"
"Uropaper A 'Eiken'" and "Uropaper B 'Eiken'"
Launched sales of immunological and serological reagents
 Hemolytic streptococcal infection testing reagent "Streptolysin O 'Eiken'"
Hemolytic streptococcal infection testing reagent "Streptolysin O 'Eiken'"
1965
Completed phase-one construction of the Nogi Plant (Nogi, Tochigi: Nogi Division) and launched production
 Completed Nogi Plant phase-one construction
Completed Nogi Plant phase-one construction
Launched sales of blood culture medium bottles
 "Culture Bottle No. 2"
"Culture Bottle No. 2"
Started the Saburo Kojima Memorial Society (business taken over by Kurozumi Medical Foundation (K M F) in 1993)
1967
Launched sales of latex agglutination immunological and serological reagents
 Rheumatoid factor measurement reagent "RA Reagent 'Eiken'"
Rheumatoid factor measurement reagent "RA Reagent 'Eiken'"
1969
Company name changed to Eiken Chemical Co., Ltd. on the 30th anniversary of its founding
 Eiken Chemical Co., Ltd. company badge
Eiken Chemical Co., Ltd. company badge
1973
Launched sales of "Pore Media" prepared culture medium
 "Pore Media"
"Pore Media"
1975
Completed Eiken Immunochemical Research Laboratory (Kita-ku, Tokyo: Oji Office) and launched operations
 Eiken Immunochemical Research Laboratory
Eiken Immunochemical Research Laboratory
Launched sales of Radioimmunoassay (RIA) reagents
Launched sales of slide plate agglutination reagents using latex agglutination reaction
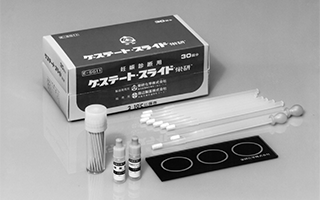 "Gestate Slide 'Eiken'"
"Gestate Slide 'Eiken'"
1977
Launched sales of bacterial identification reagents
Launched sales of the first immunological and serological reagents developed with a pioneering method that does not dilute the specimen
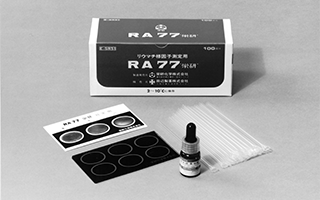 "RA77 'Eiken'"
"RA77 'Eiken'"
1980
Completed new headquarters building (Bunkyo-ku, Tokyo) and launched operations
 New headquarters building
New headquarters building
1981
Tsuyoshi Kurozumi, President, awarded the 4th Order of the Sacred Treasure for his achievements contributing to the improvement of health and hygiene both inside and outside of Japan
Launched sales of immunological and serological reagents using the SRID method
Opened the Toda Research Laboratory
1982
Launched sales of reagents related to enzyme immunoassay methods (EIA method and ELISA method)
Launched sales of specialized reagents for use with fully-automated immunochemical analyzer using a latex agglutination reaction
 "LA-SYSTEM"
"LA-SYSTEM"
1984
Completed phase-one construction for the Nasu Plant (Otawara, Tochigi: Nasu Office) and launched production
 Nasu Plant
Nasu Plant
Launched sales of urinary salt content measurement reagents
 "Saltpaper 'Eiken'"
"Saltpaper 'Eiken'"
1985
Launched sales of "KB Disk 'Eiken'" bacterial drug sensitivity testing reagents
1986
Appointed Tadao Kurozumi as President and Representative Director
 Tadao Kurozumi with Tsuyoshi
Tadao Kurozumi with Tsuyoshi
Launched sales of blood glucose self monitoring equipment and reagents
1987
Launched sales of in vivo diagnostic contrast media
 "Hexabrix 320"
"Hexabrix 320"
Launched sales of fecal immunochemical test reagents
 "OC-Hemodia"
"OC-Hemodia"
Launched sales of the "BACTSYSTEM" bacterial data management system
1988
Opened resident office in San Diego, USA
Launched sales of bacterial drug sensitivity testing reagents (fluid culture dilution method)
Launched distribution of Tosoh Corporation enzyme immunoassay equipment (AIA) and dedicated reagents
1989
Launched sales of the world's first fully-automated fecal occult blood analyzer and dedicated reagents
 "OC-SENSOR"
"OC-SENSOR"
1990
Listed stock in the Second Section of the Tokyo Stock Exchange
 January 29, 1990 Nihon Keizai Shimbun
January 29, 1990 Nihon Keizai Shimbun
1991
Launched sales of "Uropaper GP 'Eiken'" OTC urinalysis test strip
1992
Completed Biochemical Research Laboratory (Nogi, Tochigi) and launched operations
 Biochemical Research Laboratory
Biochemical Research Laboratory
Launched sales of automated urine analyzer
 "US-2100"
"US-2100"
Closed Katsushika Office and Toda Research Laboratory
1993
Established the Kurozumi Medical Foundation (K M F), presented first research grants
Launched sales of "LZ Series" reagents for clinical chemistry analyzers using the latex agglutination reaction
Launched sales of "Lumispot AL-1000" chemiluminescent immunoassay analyzer and dedicated reagents
Began overseas expansion of fecal immunochemical test reagents and analyzer business
1994
Launched sales of fecal immunochemical test reagents for manual
 "OC-Hemocatch 'Eiken'"
"OC-Hemocatch 'Eiken'"
1996
Launched sales of "US-3100" and "US-3200" fully-automated urine analyzer and "Uropaper α II 'Eiken'" dedicated reagents
1999
Announced development of LAMP method new gene amplification technology
Launched sales of "US-2100R" automated urine analyzer
2000
Launched sales of "OC-SENSOR μ(Micro)" fully-automated fecal occult blood analyzer
2001
Terminated sales agreement with Tanabe Seiyaku, began in-house sales of in vitro diagnostics and equipment (terminated capital alliance in 2007)
Launched sales of "OC-SENSOR neo" fully-automated fecal occult blood analyzer
Launched sales of "US-3100R" fully-automated urine analyzer
2002
Selected as one of the specified brands of the Tokyo Stock Exchange First Section
Launched sales of "Loopamp Series" genetic testing reagents and measurement equipment utilizing LAMP
 "Loopamp Bovine Embryo Sexing Kit"
"Loopamp Bovine Embryo Sexing Kit"
2003
Launched sales of for LAMP-based in vitro diagnostics
"Loopamp SARS-CoV detection kit"
2004
Established EIKEN SHANGHAI CO., LTD. (currently EIKEN CHINA CO., LTD.)
 EIKEN SHANGHAI CO., LTD.
EIKEN SHANGHAI CO., LTD.
2005
Acquired ISO13485 and ISO9001 certifications
Executed a joint development contract with FIND for a rapid genetic tuberculosis detection method based on the LAMP method
Launched sales of "US-1000" automated urine analyzer
2006
Executed a business partnership agreement and capital alliance with Otsuka Pharmaceutical Co., Ltd.
Launched sales of "Dry Plate 'Eiken' 192" bacterial drug sensitivity testing reagent plates
 "Dry Plate 'Eiken' 192" plates
"Dry Plate 'Eiken' 192" plates
2007
Participated in planning for the BRAVE CIRCLE campaign to eradicate colorectal cancer
Launched sales of "OC-SENSOR DIANA" fully-automated fecal occult blood analyzer
Merged with EIKEN KIZAI CO., LTD., previously a subsidiary
Launched sales of "US-3300" fully-automated urine analyzer
Appointed Tetsuya Teramoto as President and CEO
 Tetsuya Teramoto
Tetsuya Teramoto
2008
Relocated headquarters and integrated three Tokyo offices (Taito-ku, Tokyo)
Successfully developed the procedure for ultra rapid extraction method (PURE method) for use with the LAMP method
Launched sales of "US-2200" automated urine analyzer
Launched sales of "US-3100Rplus" fully-automated urine analyzer
2009
Launched sales of "LoopampEXIA (LA-500)" real-time turbidimeter for LAMP
 "LoopampEXIA"
"LoopampEXIA"
Launched sales of "OC-SENSOR io" fully-automated fecal occult blood analyzer
2010
Acquired ISO14001 certification
2011
Established Europe office in Amsterdam, Holland
2012
Completed Operation Management Center (OMC Building) at the Nogi Division and launched operations
 OMC Building
OMC Building
Launched sales of "Immunocatch Series" rapid diagnostics test kits
 "Immunocatch-Noro"
"Immunocatch-Noro"
Launched sales of fully-automated bioluminescent enzyme immunoassay analyzer and dedicated reagents
 "BLEIA-1200"
"BLEIA-1200"
2014
Appointed Morifumi Wada as President and CEO
 Morifumi Wada
Morifumi Wada
Launched sales of "OC-SENSOR PLEDIA" fully-automated fecal occult blood analyzer
 "OC-SENSOR PLEDIA"
"OC-SENSOR PLEDIA"
2015
Began development of compact, next-generation fully-automated genetic testing equipment and multi-item testing chips utilizing LAMP
Launched sales of "US-3500" fully-automated urine analyzer
 "US-3500"
"US-3500"
2016
Formed a tie-up with Sysmex Corporation for the urine chemistry testing business in overseas markets
LAMP-based tuberculosis molecular diagnostics (TB-LAMP) recommended by WHO
 WHO recommendation
WHO recommendation
Executed global distribution contracts for tuberculosis and malaria genetic diagnostic reagents and equipment use of the LAMP method through agreement with HUMAN, Germany
Completed Dried Product Plant (DPP Building) at the Nogi Division, established an expanded production line for urinalysis test strips
 DPP Building
DPP Building
Launched sales of "US-1200" automated urine analyzer
2018
Conducted preparations for expanding the Nogi Division
"Modern Media" academic information magazine reached 750 volumes
2019

2020
Launches the Loopamp 2019-nCoV Detection Kit
2021
Launched sales of automated urine analyzer "US-2300"
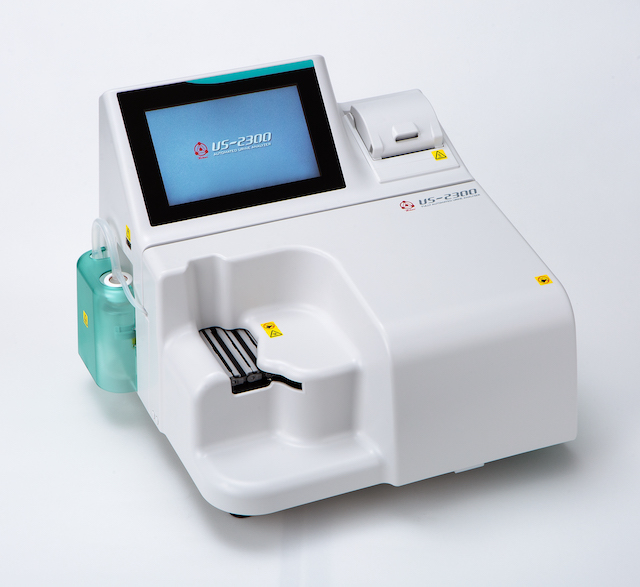 US-2300
US-2300
Launched sales of fully-automated fecal occult blood analyzer "OC-SENSOR Ceres"
 OC-SENSOR Ceres
OC-SENSOR Ceres
Appointed Tsugunori Notomi as President and CEO
 Tsugunori Notomi
Tsugunori Notomi