Completed phase-one construction of the Nogi Plant (Nogi, Tochigi: Nogi Division) and launched production




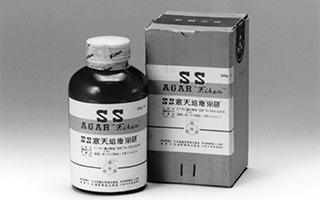
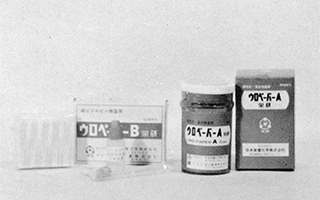
First company to succeed in creating a powdered agar for the detection of bacteria (isolation of typhoid and shigella bacteria), contributing to the development of communicable disease control and public health in Japan























"Loopamp SARS-CoV detection kit"